Esther Zanin
The Zanin lab moved to the FAU Erlangen!
Please check out new webpage here: https://www.zelldynamik.nat.fau.de
Mechanisms of cell division
During the lifetime and development of an organism millions of cell divisions occur. At the end of each cell division the mother cell physically splits into two daughter cells in a process called cytokinesis. Defects in cytokinesis can result in birth defects, miscarriage, as well as cancer. The goal of our work is to reveal the molecular mechanisms that control cell division and cytokinesis in time and space.
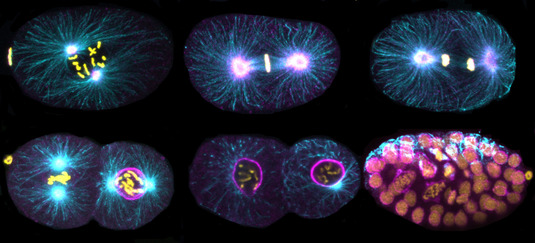
Immunofluorescence images of C. elegans embryos. DNA (yellow), Tubulin (cyan), TPXL-1 (magenta).
During cytokinesis a contractile actin-myosin ring assembles at the cell equator underneath the plasma membrane. Contractile ring formation has to be coordinated temporally with cell cycle progression and spatially with chromosome segregation. The contractile ring is placed between the segregating chromosomes by the mitotic spindle and thereby distribution of the genomic content is coordinated with the physical separation of the mother cell. The mitotic spindle promotes the activation of the GTPase RhoA in a narrow region at the cell equator. RhoA in turn induces contractile ring formation by activating myosin II and inducing the formation of filamentous actin. The aim of our work is to reveal the molecular mechanisms of how contractile ring formation is controlled in time and space. To achieve this aim we use a combination of biochemistry, genetics and quantitative live-cell microscopy in the C. elegans and human tissue culture cells in the lab.
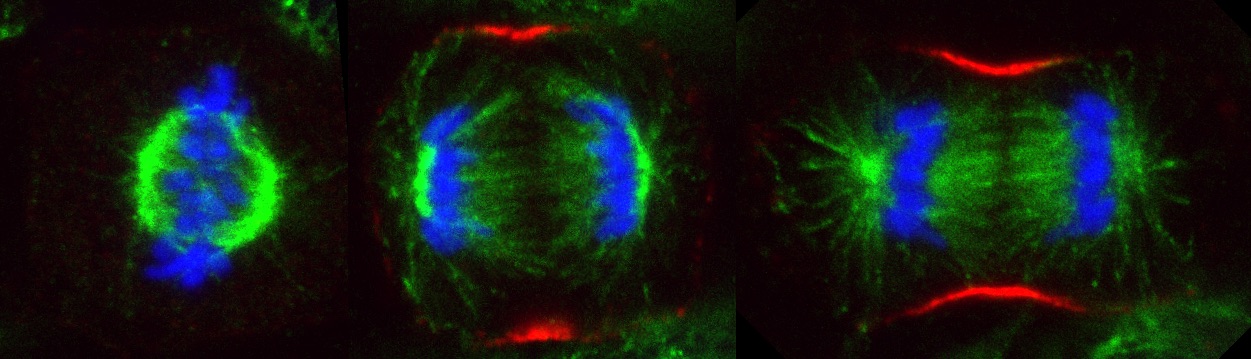
Hela cell during metaphase and anaphase. DNA (blue), Tubulin (green), Contractile ring (red)

